- ++66 2902 6106-8
- [email protected]
- 56/12-15 Phrayasuren 45,Bangkok
- Home
- Our Story
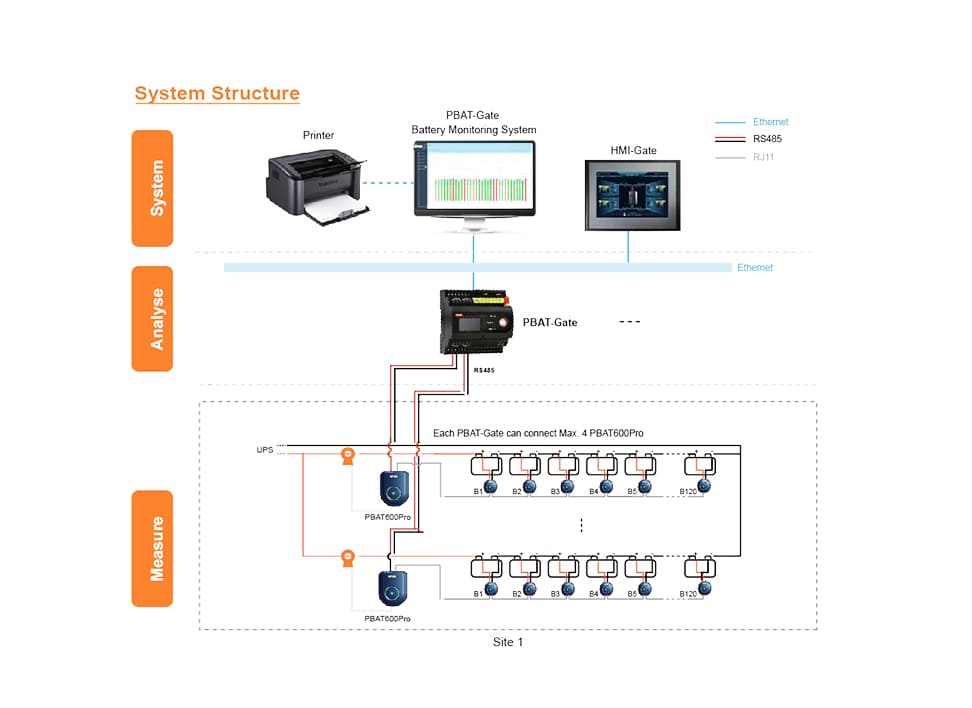
- Product
Uninterruptible Power Supply
Line interactive
Pure Sine Wave
Uninterruptible Power Supply
Online UPS [ 1:1]
Online Transformer-Based UPS [3:3]
Modular UPS [3:3]
Relay Type 1 Phase
Servo motor type 1 phase
Servo motor type 3 phase
- Services
- Contact Us
- Product Register